COVID-19 vaccines are used to prepare the body's immune system to protect against COVID-19.
Everyone ages 6 months and older should get a 2025-2026 COVID-19 vaccine. This includes pregnant women and those planning to become pregnant. You should get a 2025-2026 COVID-19 vaccine even if you:
- Already had COVID-19
- Have been vaccinated with earlier versions of the vaccine
Information
HOW COVID-19 VACCINES WORK
COVID-19 vaccines protect people from getting COVID-19 and from getting more severe symptoms if they do get COVID-19. These vaccines teach your body how to defend against the SARS-CoV-2 virus, which causes COVID-19.
COVID-19 vaccines have been shown to do a very good job of:
- Preventing infection with the SARS-CoV-2 virus, which causes COVID-19
- Protecting against serious illness, hospitalization, and death from COVID-19
- Reducing the risk of people spreading COVID-19
mRNA VACCINES
The mRNA vaccines approved in the United States work differently from many other vaccines.
- COVID-19 mRNA vaccines use messenger RNA (mRNA) to tell cells in the body how to briefly create a harmless piece of spike protein that is unique to the SARS-CoV-2 virus. Cells then get rid of the mRNA.
- This spike protein triggers an immune response inside your body, making antibodies that protect against COVID-19. Your immune system then learns to attack the SARS-CoV-2 virus if you are ever exposed to it.
There are 3 mRNA COVID-19 vaccines currently approved for use in the United States:
- The Pfizer-BioNTech vaccine is approved for use in people age 5 years and older.
- There are 2 Moderna COVID-19 vaccines, one for people ages 6 months and older and another for people 12 years and older.
The COVID-19 mRNA vaccine is given as an injection (shot) in the arm. The 2025-2026 mRNA vaccines protect against the current strains of the COVID-19 virus.
SUBUNIT VACCINE
The Novavax vaccine is a protein subunit vaccine. The vaccine includes harmless pieces of the spike protein of the SARS-CoV-2 virus. The vaccine triggers the body to develop antibodies to protect you from the virus. The 2025-2026 Novavax vaccine also protects against current strains of the COVID-19 virus. It is approved for use in people ages 12 years and older.
VACCINATION SCHEDULE
The vaccination schedule is based on:
- Your age
- Whether you are moderately or severely immunocompromised
- Whether you have recently had COVID-19 (within 3 months)
The Centers for Disease Control and Prevention recommend that people of all ages consult their health care provider to decide if getting a COVID-19 vaccine is best for them. The provider may be a doctor, nurse, or pharmacist. This process is called shared decision-making.
Most health experts recommend:
- Everyone ages 6 months and older should get 1 dose of the 2025-2026 COVID-19 vaccine.
- People with at least one underlying health condition that puts them at high risk for severe outcomes from COVID-19 should get 1 dose of the 2025-2026 COVID-19 vaccine. The Centers for Disease Control and Prevention provides a list of health conditions and COVID-19 risk factors that increase the risk for severe disease.
- Women who are pregnant, thinking about becoming pregnant, or breastfeeding should receive 1 dose of the 2025-2026 COVID-19 vaccine.
Adults and children who are severely immunocompromised may need additional doses of the 2025-2026 COVID-19 vaccine.
Talk with your health care provider about whether you may need additional doses.
People who recently had COVID-19 may delay getting a COVID-19 vaccine for 3 months. You are much less likely to get COVID-19 in the 3 months after having the illness. You may choose to get the vaccine sooner if you or someone in your family are at severe risk of illness or if there are high local rates of COVID-19.
VACCINE MYTHS
COVID-19 vaccines:
- Do not contain any live virus, and they cannot give you COVID-19
- Do not affect or interfere with your genes (DNA)
- Do not affect or interfere with pregnancy, nor do they make you infertile
VACCINE SIDE EFFECTS
While COVID-19 vaccines will not make you sick from COVID-19, they may cause certain side effects and flu-like symptoms. This is normal. These symptoms are a sign that your body is making antibodies against the virus.
Side effects can vary from person to person. Common side effects include:
- Pain, redness, or swelling on the arm where you got the shot
- Tiredness
- Headache
- Muscle pain
- Chills
- Fever
- Nausea
- Swollen lymph nodes
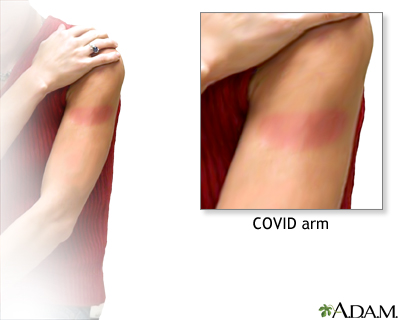
Some side effects from the shot may affect your ability to do daily activities, but any side effects will go away in a few days. Any side effects from the vaccine are far less dangerous than the potential for serious illness or death from COVID-19.
HOW TO GET THE VACCINE
There are several ways you can look for vaccination providers near you.
- Ask your health care provider if they offer COVID-19 vaccines.
- Check your local pharmacy's website or call the pharmacy to see if vaccination appointments are available.
- Contact your state or local health department to find additional vaccination locations in your area.
Learn what to expect when you get your COVID-19 vaccine.
VACCINE SAFETY
The safety of vaccines is the top priority, and COVID-19 vaccines have passed rigorous safety standards before approval. Millions of people have received the vaccine, and no long-term side effects have been reported. The vaccines continue to be closely monitored to ensure they are safe and effective.
There have been reports of some people who have had an allergic reaction to the current vaccines. So it is important to follow certain precautions:
- If you have ever had a severe allergic reaction (called anaphylaxis) to a particular type of COVID-19 vaccine, you should not get that vaccine. You may be able to get another type of COVID-19 vaccine.
- If you have a non-severe allergic reaction after getting the first shot of the COVID-19 vaccine, you may be able to get another dose in some cases. Your provider may refer you to an allergy and immunology provider for care before considering getting the vaccine.
- If you only had a skin rash on the arm in which you got the shot (COVID-19 arm), you may still get additional shots.
If you have had an allergic reaction, even if not severe, to other vaccines or injectable therapies, you should ask your provider if you should get a COVID-19 vaccine. Your provider will help you decide if it is safe to get vaccinated.
Serious health events from COVID-19 vaccines, such as an allergic reaction, are rare. Adverse events after COVID-19 vaccination are very rare.
Rare cases of myocarditis (inflammation of the heart muscle) and pericarditis (inflammation of the outer lining of the heart) have been reported in children and teens ages 5 years and older after getting the COVID-19 vaccine.
This reaction occurred more often in male adolescents and young adults ages 12 to 39 years. However, it has also occurred in females, in other age groups, after other doses, and after receiving any type of vaccine.
- It occurs more often after getting a second dose, most often within 7 days after vaccination. Studies show that this rare risk may be reduced by waiting 8 weeks between the first and second dose.
- With proper care and rest, most people who had the reaction got better quickly without any lasting effects.
- For people who had this rare reaction, it is important to talk with a cardiologist (heart doctor) about how and when to return to exercise and sports.
Symptoms of myocarditis and pericarditis include:
- Chest pain
- Shortness of breath
- Fast-beating heart, fluttering, or pounding heart
If your child or teenager has any of these symptoms, get medical help right away.
All these associations are so rare that they should not cause hesitation in receiving any of these vaccines.
The CDC recommends that people may still get vaccinated if they have a history of:
- Severe allergic reactions not related to vaccines or injectable medicines -- such as food, pet, venom, environmental, or latex allergies
- Allergies to oral medicines or a family history of severe allergic reactions
Alternative Names
Vaccines for COVID-19; COVID-19 vaccinations; COVID-19 shots; Vaccinations for COVID-19; COVID-19 immunizations; COVID-19 prevention - vaccines; mRNA vaccine - COVID-19; COVID-19 vaccine booster shots; Booster shots for COVID-19
References
Centers for Disease Control and Prevention website. Vaccine safety: coronavirus disease 2019 (COVID-19) vaccine safety. www.cdc.gov/vaccine-safety/vaccines/covid-19.html. Updated January 31, 2025. Accessed October 22, 2025.
Centers for Disease Control and Prevention website. COVID-19: benefits of getting vaccinated. www.cdc.gov/covid/vaccines/benefits.html. Updated June 11, 2025. Accessed October 22, 2025.
Committee on Infectious Diseases. Recommendations for COVID-19 Vaccines in Infants, Children, and Adolescents: Policy Statement. Pediatrics. 2025 Aug 19. Epub ahead of print. PMID: 40826495. pubmed.ncbi.nlm.nih.gov/40826495/.
Prasad V, Makary MA. An Evidence-Based Approach to Covid-19 Vaccination. N Engl J Med. 2025 Jun 26;392(24):2484-2486. Epub 2025 May 20. PMID: 40392534. pubmed.ncbi.nlm.nih.gov/40392534/.
The American College of Obstetricians and Gynecologists website. COVID-19 Vaccination Considerations for Obstetric–Gynecologic Care. www.acog.org/clinical/clinical-guidance/practice-advisory/articles/2020/12/covid-19-vaccination-considerations-for-obstetric-gynecologic-care. Updated September 2025. Accessed October 22, 2025.
US Food and Drug Administration website. COMIRNATY. www.fda.gov/vaccines-blood-biologics/comirnaty. Updated September 25, 2025. Accessed October 22, 2025.
US Food and Drug Administration website. MNEXSPIKE. www.fda.gov/vaccines-blood-biologics/mnexspike. Updated September 25, 2025. Accessed October 22, 2025.
US Food and Drug Administration website. NUVAXOVID. www.fda.gov/vaccines-blood-biologics/vaccines/nuvaxovid. Updated September 25, 2025. Accessed October 22, 2025.
US Food and Drug Administration website. SPIKEVAX. www.fda.gov/vaccines-blood-biologics/spikevax. Updated September 25, 2025. Accessed October 22, 2025.
Review Date 1/1/2025
Updated by: Linda J. Vorvick, MD, Clinical Professor Emeritus, Department of Family Medicine, UW Medicine, School of Medicine, University of Washington, Seattle, WA. Also reviewed by David C. Dugdale, MD, Medical Director, Brenda Conaway, Editorial Director, and the A.D.A.M. Editorial team. Editorial update 10/23/2025.